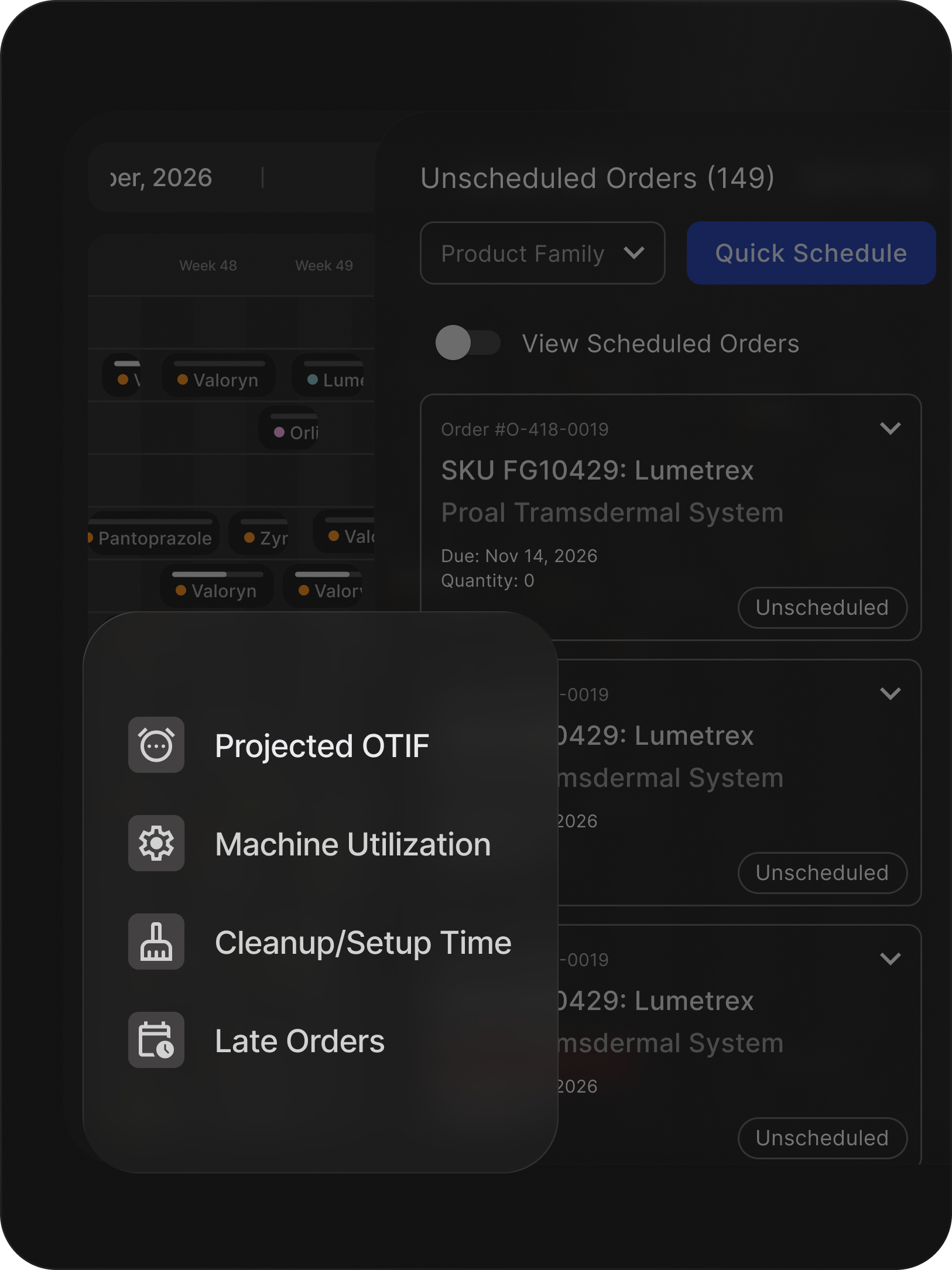
TLDR:
Pharma shop floor scheduling software improves production efficiency by turning static plans into executable, constraint-aware schedules. It embeds real-world factors like equipment availability, validation rules, labor qualifications, and material readiness directly into daily sequencing.
This enables faster response to disruptions, better resource utilization, and stronger compliance. As complexity grows, it replaces manual spreadsheet coordination with structured, real-time scheduling that stabilizes production and reduces variability.
How Pharma Shop Floor Scheduling Software Improves Production Efficiency
Production efficiency in pharmaceutical manufacturing rarely breaks down because capacity is insufficient. It breaks down when daily execution falls out of sync.
Each day, the following variables must align:
-
Equipment availability
-
Labor qualifications
-
Validation rules
-
Demand priorities
That alignment does not happen on its own. Production teams must actively manage it. Without that structure, inefficiencies build quickly across the production floor.
As product portfolios expand and regulatory scrutiny tightens, manual scheduling methods struggle to keep pace. Variability rises. Changeovers multiply. Small disruptions demand outsized effort.
At that point, the issue is no longer how hard teams are working. It is whether the scheduling system itself can sustain coordinated execution at scale.
Strengthening Execution Discipline on the Shop Floor
Production efficiency depends on whether daily schedules reflect real operating conditions rather than ideal assumptions. Weekly plans built in isolation rarely withstand the variability of live production.
Equipment conflicts arise, window-cleaning overlaps, material readiness shifts, and static schedules degrade quickly under operational pressure.
This shift applies sequencing logic to live production data, validated equipment rules, and capacity interdependencies.
Instead of relying on manual intervention, the system embeds production logic directly into the scheduling framework. Three structural shifts typically follow.
From Static Planning to Executable Sequencing
Traditional planning tools assume predictable cycle times and uninterrupted availability. In pharmaceutical manufacturing, batch execution involves:
-
Cleaning validation windows
-
Quality hold periods
-
Shared equipment dependencies
-
Operator qualification limits
-
Batch size variability
-
Material release timing
Modeling these variables directly within the schedule transforms planning from theoretical alignment into executable sequencing. The schedule reflects operational reality rather than managerial intent.
From Lagging Reports to Real-Time Execution Insight
Real-time dashboards are often presented as a speed advantage. In pharma, control matters more than speed.
Delayed visibility increases compliance exposure and production instability. Access to current data on work-in-progress, utilization, and sequencing deviations allows supervisors to intervene before small disruptions expand.
The operational effect is a shift from reactive correction to managed execution.
From Resource Conflict to Balanced Utilization
Mid-market pharmaceutical facilities often operate with limited high-value assets. Compression lines, coating systems, and packaging suites serve multiple products under strict validation constraints.
Uncoordinated sequencing creates uneven workload distribution. Some assets are idle while others are overloaded.
Systematic sequencing discipline reduces avoidable cleaning cycles by deliberately grouping batches. Labor capacity aligns more closely with equipment demand.
Throughput becomes predictable rather than volatile. The result is not merely higher output. It is a stable performance across constrained resources.
How Shop Floor Scheduling Software Improves Production Efficiency During Disruptions
Disruption is inherent to manufacturing. Equipment failures, material delays, and extended investigations occur regularly. Efficiency depends on how rapidly the schedule absorbs these events.
In manual environments, each event triggers cascading adjustments. Planners reassess capacity, verify sequencing dependencies, confirm labor availability, and rebuild timelines.
During this period, idle time increases and variability compounds. With scheduling logic embedded in the system, recalculation occurs against predefined rules.
Alternatives are evaluated based on:
-
Equipment availability
-
Batch priority
-
Validated sequencing constraints
-
Labor qualifications
-
Material readiness
Scenario modeling lets leaders compare outcomes before changing the schedule. This reduces instability during periods of stress.
For mid-market manufacturers operating without excess capacity buffers, this responsiveness protects downstream packaging, distribution, and customer commitments. In this context, agility becomes structured rather than improvised.
How Shop Floor Scheduling Software Supports Regulatory Compliance
Compliance in pharmaceutical manufacturing is not separate from daily operations. It runs through them.
Every batch sequence involves equipment assignments, cleaning validation windows, operator qualifications, and quality release checkpoints.
When these are managed manually, the documentation trail can become inconsistent. Entries get delayed. The executed sequence may not match what was recorded in the plan.
Scheduling software addresses this at the execution layer. Validated equipment assignments, approved cleaning sequences, and qualification-based operator allocations are built directly into the scheduling logic.
A batch cannot be assigned to an asset that has not cleared its validation window. An operator cannot be scheduled for a task outside their scope of certification.
This reduces the risk of procedural deviation before it reaches the shop floor rather than catching it afterward.
Aligning Production With ERP and Supply Chain Systems
Scheduling decisions influence procurement timing, inventory levels, quality release schedules, and customer delivery commitments. When execution tools operate separately from ERP systems, misalignment becomes routine.
Enterprise resource planning systems manage demand aggregation, purchasing, and financial reporting. They typically do not govern detailed batch sequencing across shared assets.
This separation introduces friction. Orders released based on forecast demand may conflict with equipment constraints or validation sequences on the production floor. Manual reconciliation follows.
By linking order data from ERP to execution-level scheduling logic, production plans reflect real capacity conditions. Delivery forecasts become grounded in operational feasibility.
Measuring the Impact on Production Efficiency
These changes must translate into measurable performance. In pharmaceutical environments, production efficiency is reflected through reliability and asset productivity rather than raw output volume.
When sequencing discipline and real-time coordination are embedded into daily execution, several indicators stabilize.
Schedule adherence
Adherence measures the degree to which actual production follows planned sequencing. In spreadsheet-driven environments, deviation increases as manual adjustments accumulate.
When schedules reflect real constraints, adherence stabilizes. Downstream variability decreases.
Equipment utilization
Validated production assets represent a significant capital investment. Idle capacity directly affects margin performance.
The practical consequence is fewer idle windows and more evenly distributed workloads across shifts. Utilization improves not through acceleration, but through predictability.
Over time, these operational improvements influence capital deployment decisions.
More predictable equipment utilization and stabilized throughput allow manufacturers to defer capacity expansion and avoid premature capital expenditure.
Changeover efficiency
Changeovers in pharmaceutical manufacturing include cleaning verification, documentation, and quality confirmation.
These transitions are necessary but time-intensive. Logical batch grouping reduces avoidable product switches where validation rules permit. Recovered hours accumulate across the production calendar.
On-time delivery performance
Internal sequencing reliability shapes external customer commitments. When batch completion dates fluctuate, distribution timelines follow.
The effect is fewer last-minute sequencing changes. Delivery performance becomes consistent rather than optimistic.
Planning overhead
Manual schedule reconstruction consumes managerial capacity. When recalculation and dependency modeling are automated, planners evaluate trade-offs instead of rebuilding spreadsheets.
Taken together, these indicators signal a broader shift from reactive coordination to governed operational performance.
Why Spreadsheet Scheduling Fails as Complexity Scales
Spreadsheets function adequately in stable, low-variability environments. Many mid-market pharmaceutical companies begin with Excel because it offers flexibility and familiarity.
Complexity changes that equation.
Each additional SKU introduces new sequencing dependencies. Cleaning windows expand. Equipment sharing intensifies. Documentation requirements multiply. Planners struggle to model these interdependencies manually.
Spreadsheets do not dynamically account for:
-
Shared equipment constraints
-
Validation sequencing rules
-
Operator certification limits
-
Batch size variation
-
Material readiness
-
Shift-based capacity restrictions
As these variables increase, decision latency grows. Conflicts are identified after schedules are already in motion.
Risk accumulates quietly, and a single outdated file version or overlooked dependency can cascade into production delay and compliance exposure.
At scale, the constraint is not planner capability. It is an architectural limitation.
Comparing Manual Scheduling to Structured Scheduling Software
The differences between spreadsheet-based scheduling and dedicated shop floor scheduling software are most visible under conditions of complexity and disruption.
In stable, low-variability environments, the gap is manageable. As product portfolios grow and manufacturing processes become more interdependent, the gap widens. The table below outlines how each approach handles common operational challenges in pharmaceutical manufacturing:

This table reflects where those gaps typically show up first: in conflict detection, disruption response, and compliance documentation.
Sustaining Production Efficiency as Complexity Increases
Production efficiency in pharmaceutical manufacturing is shaped by the discipline embedded in daily coordination. As product portfolios expand and regulatory demands intensify, sequencing decisions carry increasing operational weight.
How does shop floor scheduling software improve production efficiency? It strengthens the decision architecture that governs resource allocation, execution alignment, and disruption response.
Mid-market pharmaceutical companies reach a clear inflection point. When complexity outpaces manual coordination tools, variability follows.
Sustained efficiency demands systems that manage interdependencies deliberately rather than informally.
Organizations that formalize this capability early convert operational discipline into durable competitive stability.
Frequently Asked Questions
What is shop floor scheduling software in pharmaceutical manufacturing?
Shop floor scheduling software governs daily batch sequencing by aligning equipment, labor, validation requirements, and demand priorities within regulated manufacturing environments.
How does shop floor scheduling software improve production efficiency?
It improves production efficiency by embedding sequencing logic, resource constraints, and real-time production data into the scheduling process, reducing variability and increasing asset productivity.
Can shop floor scheduling software integrate with an ERP system?
Yes. It connects execution-level scheduling with enterprise resource planning systems to align demand signals, inventory data, and production timelines.
Why do spreadsheets become unreliable for pharmaceutical scheduling?
Spreadsheets cannot dynamically model equipment interdependencies, validation rules, and capacity constraints at scale, which increases manual workload and operational risk.
Does scheduling software support regulatory compliance?
Yes. Advanced systems incorporate validated equipment assignments, cleaning sequences, and qualification requirements directly into scheduling logic.
Is this suitable for mid-market pharmaceutical companies?
Yes. Structured scheduling platforms are particularly valuable for manufacturers that have outgrown spreadsheets but do not require complex enterprise-wide systems.
